MULTISCALE DYNAMICS
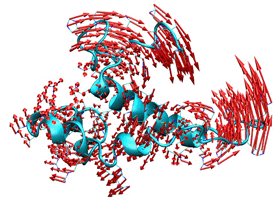
We actively work to effectively address the study and simulation of the dynamics of biomolecular systems with Normal-Mode Analysis (NMA), geometric algebra, and other multiscale approximations. The structural flexibility of biomolecules is closely coupled to function, as evidenced by many conformational changes observed on key cellular processes. Following the basic principle if you know how it moves, you can infer how it works; inferring the intrinsic molecular flexibility from a single conformation could offer a direct link to understand functional motions. We address this problem by exploring novel multiresolution approaches to simulate the dynamics of protein, nucleic acids, and their complexes. If you are interested you can play with iMOD, our versatile toolkit to perform Normal Mode Analysis (NMA) in internal coordinates or directly in our iMODS online server. To explore efficiently local protein loop conformations, we add constraints inside our NMA formulation to maintain the position and orientation of the loop ends. This novel method using, called ilMODE, compared with other concerted ways to move loops i) have no size limitations 2) It is very efficient since it only requires the diagonalization of a tiny matrix of size 2N-5, being N the number of residues of the loop iii) the elastic network includes both the loop and protein neighbor residues.